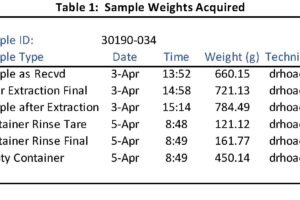
- Particulate is defined by the method used to measure it. Unlike chemically distinct emissions like carbon monoxide or nitrogen oxide, “particulate” can be composed of many different distinct compounds. Some of these compounds may be volatile and may be “particulate” at lower temperatures but gaseous at higher temperatures.
- US EPA Method 5. This test method isokinetically extracts a sample from the stack or duct and passes that sample through a filter. The sample probe and filter are heated to 248 °F. Any particulate captured on the filter is (logically) referred to as “filterable particulate” or FPM; that is, any material not gaseous at that temperature is captured. There are variants of Method 5 (for example, 5B and 5F) that use other temperatures for sample collection.
- US EPA Method 202. The gaseous material sampled with Method 5 passes through the filter and a temperature-controlled condenser followed by two, initially dry, impingers – essentially big test tubes. As the gas is cooled, water and other compounds (both organic and inorganic) condense out. Any material collected in these impingers (minus the water) is referred to as “condensable particulate” or CPM. The logic used for this method is that any condensed material would also condense and form particulate when the gases exit the stack and are cooled in the ambient air. More on this assumption in a moment…
- Method 202 Bias Issues. Unfortunately, there are often other compounds present in the sample gas that may react when placed together in a large, wet impinger. Two compounds commonly found in some gas streams are ammonia (NH3) and sulfur dioxide (SO2). Left on their own in the stack, these two gases will not normally combine even upon exiting the stack. However, when mixed together in an impinger with even a small amount of water, the two gases combine to form ammonium salts – a particulate. The formation of ammonium salts is an artifact of Method 202 sampling and does not occur with gas exiting the stack. Therefore, Method 202, can be biased high for gases streams containing these two compounds — the method creates “false particulate.”
- A Synergistic Effect. NH3is highly water soluble. During a test, as NH3is dissolved in the impinger water, the pH of the water is raised. The higher pH enhances the collection of SO2. — in effect, a little caustic scrubber is created. The higher the pH, the more efficiently SO2is scrubbed. The more NH3and SO2are captured, the higher the Method 202 bias. Some may say that when Method 202 was modified a few years back, it fixed the bias problem. Unfortunately, there is ample evidence that while the bias issues are better than they were with the old method, they are still with us. The addition of the “dry” impingers and CPM filter in the new method helped but did not completely solve the bias problem.
- What to Do? For facilities where highly biased CPM results are an issue, here is an approach that can work. For Method 202 analysis, results are split into organic and inorganic fractions. It is the inorganic fraction that typically is biased. It is where the ammonium salts are found. So the solution? Use only the organic fraction. But wait, I hear you say. What about any other inorganic condensable compounds that may be legitimately considered particulates? We can’t just throw away all the inorganics, can we?
- Controlled Condensation. In any gas stream that contains SO2, particularly those with catalysts to control NOx, there will be some SO3/H2SO4– sulfuric acid. At temperatures well above ambient, sulfuric acid will form a mist which is legitimately considered a particulate. At Method 5 collection temperatures (248 °F) Much of this mist will be captured on the filter, but some of it will make it through the filter and condense in the impingers. For many gas streams containing SO2, SO3will be the only significant contributor to CPM. This SO3must be accounted for since it is a legit particulate. The answer is to do a test that is specific to sulfuric acid mist… controlled condensation (CCM).
- Why Controlled Condensation? The CCM method is designed to condense SO3/H2SO4while minimizing water condensation. Method 202 condenses both water and acid gas simultaneously leading to the chemical interactions described above. While CCM does not completely eliminate the potential for the formation of false particulate, it performs much better than M202 in this regard.
- An unbiased CPM measurement. So a less biased measurement of CPM can be obtained by using the results of the CCM test in place of the Method 202 inorganic fraction. The total CPM would then be:
CPM = Method 202 organic fraction + CCM results
Both tests should be conducted either simultaneously or as close as possible in time. In order to use this approach, regulatory authority permission must typically be granted.
CleanAir has a white paper available on this issue with more detailed information, data, and a case study. If you are interested in this approach to unbiased CPM measurement, we can support you with research, data, and testing. Let us know if we can help.